By Sharrmila Palanyandy
The first COVID-19 cases were reported in Wuhan, China in late 2019. The Director-General of the World Health Organization (WHO) declared the outbreak of COVID-19 to be a Public Health Emergency of International Concern on 30 January 2020 and characterized it as a pandemic on 11 March 2020. The COVID-19 pandemic remains a nightmare as it has rapidly spread across the globe and has caused a high number of fatalities. Not forgetting the impact on the global economy and from which many big companies are still struggling to date.
In the wake of the pandemic, science has never witnessed a quicker development of potent vaccines. The SARS-CoV 2, Pfizer/BioNTech’s vaccine received FDA approval on 11 December 2020 and EMA approval on 21 December 2020 which is within one year of pandemic. Apart from the Pfizer/BioNTech vaccine, many other COVID-19 vaccines were developed subsequently i.e., AstraZeneca, Jannsen, Cansino, Moderna, Curevac, and etc.
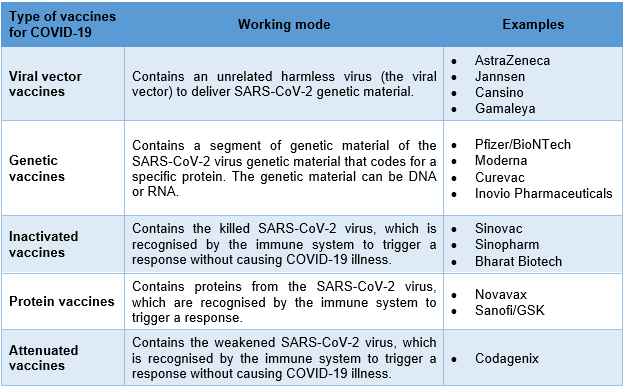
The table below summarizes the examples of COVID-19 vaccines and their working mode:
Certainly, it costs a fortune for vaccine developers to spur the new ideas and come out with the vaccine products particularly during a pandemic. Therefore, there was unrest amongst the vaccine developers when the World Trade Organization was considering to temporarily waive its 1995 agreement that would have protected intellectual property (IP) for COVID-19 vaccines and treatments. If IP is temporarily waived during the pandemic, would pharmaceutical companies stop innovating and slow down the medical progress? Let’s discuss this further.
The argument for waiving IP is stronger during the COVID-19 pandemic, as the costs of vaccine research and development were substantially offset by governments. This is because the governments poured billions into procuring raw materials, financing clinical trials, and retrofitting factories for drug companies. Further, it is known that in many circumstances the profits of large pharmaceutical companies are substantially higher than those of other (non-pharmaceutical) large companies.
Meanwhile, an argument against temporary waivers for COVID-19 vaccines was that drug companies should own the product of their labour. Are the drugs solely owned by drug companies? Beyond drug companies and governments, there are immeasurable contributions others make. Vaccines are in fact the final, translational part of product development that is years in the making which involves enormous upstream investment. Hence, it is obvious that waiving patents is not enough. But many believe that removing the threat of being sued or prosecuted would help break the logjam. It will create favourable conditions for investing in and encouraging the knowledge and technology transfer to low- and middle-income countries which need to expand their drug manufacturing capacity.
Simply put, as we are all aware, science is a social product. When Salk came up with the vaccine to eradicate polio and released it to the world in 1955, newsman Edward R. Murrow asked him whom the patent belonged to. “The people, I would say. There is no patent. Could you patent the sun?“. Salk refused to obtain patent rights simply because he wanted the global health crisis of polio to be abated.
It is obvious that, invention and thought do not occur in a vacuum but depend on the thoughts and ideas of those who came before. Since COVID-19 will not be the last pandemic humanity faces, expanding vaccine manufacturing is vital preparation for future pandemics. It is believed that increasing the number of manufacturers globally would not only increase supply and reduce prices of the drugs. This will provide low- and middle-income countries easy access to the drugs which can directly minimize the number of fatal cases caused due to any future pandemics.
- Budding Innovations: Patent Trends in Thailand’s Medical Cannabis Sector - April 20, 2024
- From Venice with Love and Litigation: The Cipriani Brand Dispute - March 26, 2024
- The Impact of Unlawful Franchise Agreements: SP Multitech Intelligent Homes Sdn Bhd v Home Sdn Bhd [2010] MLJU 1845 - March 25, 2024