By P. Kandiah and Geetha Kandiah
Moderna Inc., Sanofi, AstraZeneca plc, Roche, Gilead Sciences and Novartis are just some of the many pharmaceutical companies working to fast-track the development of a coronavirus vaccine. Many companies and research institutions from various countries are racing to develop the vaccine for COVID-19.
1. Who is leading the race to discover the vaccine and treatment for COVID-19?
At the time of writing, we do not know exactly. There are several companies, as well as universities and research institutions around the world all simultaneously conducting research either individually or in collaboration with each other in obtaining a vaccine, or a therapeutic drug to treat infected COVID-19 patients.
In fact, the World Health Organization (WHO) released a public statement on 13th April 2020 informing the public of their efforts to bring parties together to collaborate on COVID-19 vaccine development. More than 120 parties – scientists, physicians, funders and manufacturers – who are researching and working on the vaccine, “are brought together” as part of an international collaboration, coordinated by WHO, to help speed the availability of a vaccine against COVID-19.
Data from early-April (see below) indicated the countries that were in the race to develop a vaccine for COVID-19, with the US ahead of the pack.
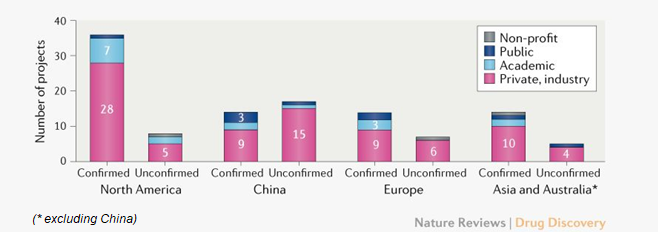 [Source: Nature Reviews Drug Discovery, https://www.nature.com/articles/d41573-020-00073-5]
[Source: Nature Reviews Drug Discovery, https://www.nature.com/articles/d41573-020-00073-5]
While pharmaceutical companies and research institutes in China were quick to embark on finding a vaccine, their progress seems to have hit a snag as the lack of new and active cases in the country means they do not have enough patients to conduct human trials and tests for the potential vaccines, putting them at a disadvantage in advancing their vaccine and treatment research.
On a related note, as of May 4th, the European Commission’s Coronavirus Global Response initiative had raised USD 8 billion towards efforts in developing vaccines, diagnostics and treatments against COVID-19 and enabling them “to be made available globally for an affordable price, regardless of where they were developed”. Interestingly, while over 30 countries, alongside the UN and research and philanthropic organizations pledged their part, the governments of the US, India and Russia did not participate, alluding perhaps to significant funding going into their own research efforts.
2. Who will own the exclusive right to manufacture the vaccine or the therapeutic drug?
The right will belong to the entity that obtains a patent right. The party that is first to file the patent will own rights to the manufacture and sale of the drugs.
As patent rights are territorial, they must file it in specific countries to gain right in that country. If it is not filed in that country, generic manufacturers can manufacture the vaccine without paying any royalty to the patent owners
3. Can more than one entity own the right?
Yes, if the invention is developed jointly by more than one entity, then all the entities can own the patent right.
However, they may agree among themselves on their separate/individual rights. For example, if a university is a joint patentee, it may not be interested to engage in commercial production and sale of the vaccine and drugs. It could, instead, either assign its rights to a pharma company or enter into a patent license with a pharma licensee.
4. There is a common belief that patented drugs are expensive and many countries, especially developing ones cannot afford the expensive vaccine or drugs. How will poor countries manage?
Patent rights are monopolistic rights. It gives rights to the party that invested in developing the drugs. That is how pharma companies recoup their investments.
But, there are times – urgent times – wherein a balance must be struck, between making a large profit and taking care of the health of the public.
Compulsory licenses are public health safeguards, included in international agreements and national laws.
Most countries have a provision in their patent laws that grant the government of the country the right to grant compulsory licences to a third party to manufacture the drug or export the drug from another country for distribution or sales (at an affordable price) to its citizens without the consent of the patentee. There will still be a royalty paid to the patent holder but at a reasonable, affordable price.
This provision is only invoked for national emergencies.
5. The Government of any country can do that? But isn’t that unjust to the inventor of the drug? A lot of expenses would have been spent in developing the drug.
It may be unjust – and for that reason, there are conditions to be met, before a country can give a compulsory licence:
- There must be no production of the patented product or application of the patented process in Malaysia without any legitimate reason;
- Or there are some but they are sold at unreasonably high prices or do not meet public demand;
- The person making the application has made efforts to obtain authorization from the owner of the patent on reasonable commercial terms and conditions but such efforts have not been successful within a reasonable period of time.
There is a limited scope to exercise the patent license and tight time limit too.
6. Has Malaysia made use of the provisions on compulsory licenses before?
Yes, Malaysia has used the provision before. In 2003, Malaysia became the first country to issue a compulsory licence for an HIV drug, and many countries followed suit.
Then, in 2017, our Government issued a compulsory license on the key hepatitis C drug, sofosbuvir. With this, Gilead’s drug could be mass produced at an affordable rate. Our government could then afford treatment for the estimated 450,000 people living with hepatitis C in Malaysia.
7. If a drug has been used before, say for example the malaria drug that is now used to treat patients with COVID-19, can any company obtain patent rights for this new treatment?
Generally, it is possible to obtain new patent rights if a known drug is now known to have a second medical use.
In fact, much of the research being done uses current existing antiviral drugs. Apart from the anti-malarial drug, the other popular one is remdesivir by Gilead. This antiviral drug, was originally developed to treat Ebola and recently US and Japan have rushed to increase production of these drugs.
If the drug can be repurposed or modified to treat COVID-19, then it may be possible to obtain patent rights of the new medical use of the known drug.
8. If so, how long will one granted patent last?
The duration of the patent will be 20 years from the date of filing the patent application.
9. Why is there a rush to obtain patent rights for the vaccine or a therapeutic drug?
A vaccine is needed for every human being on earth. That is a huge market and demand. Even if a profit of RM 1.50 for each dose is made, imagine the total sum of profit. It would be in the billions. It would be a blockbuster sale. That is why.
Only one patent will be granted in a country for the same invention. The first company to file the patent for the successful formulation of the vaccine or therapeutic drug will obtain a patent to the exclusion of all others who have been working on the same formulation.
In the latter case, they may not be able to recoup their R&D costs. And so, the stake to rush to file a patent is very high especially for a vaccine or therapeutic drug for COVID- 19.
[Our interview on this topic first appeared on Bernama TV’s “The Nation”: www.bernama.com/tv. Watch it on www.youtube.com/KASStheIPexperts]
- Budding Innovations: Patent Trends in Thailand’s Medical Cannabis Sector - April 20, 2024
- From Venice with Love and Litigation: The Cipriani Brand Dispute - March 26, 2024
- The Impact of Unlawful Franchise Agreements: SP Multitech Intelligent Homes Sdn Bhd v Home Sdn Bhd [2010] MLJU 1845 - March 25, 2024